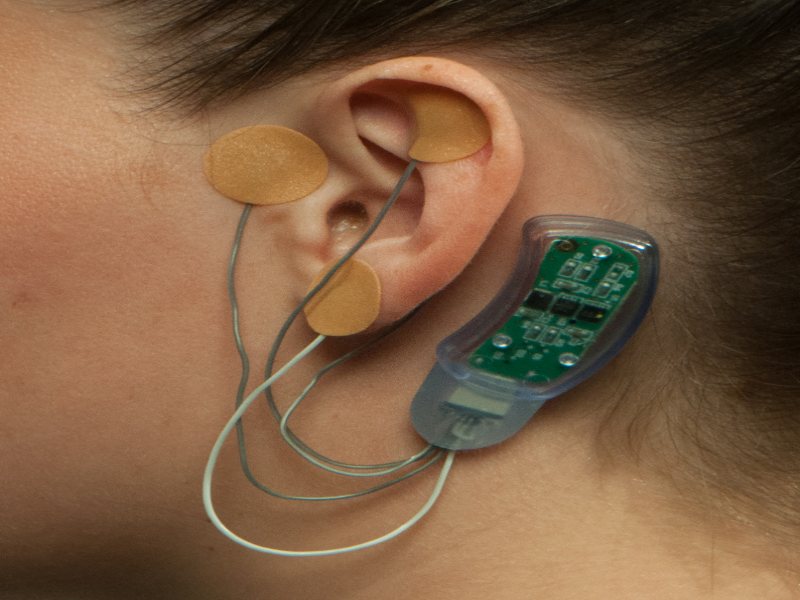
Behind The Ear Wearable Device For Opioid Treatment; California-based company Proteus, maker of a behind the ear wearable device for opioid treatment, has received FDA approval for its new product.
The device sends information to a smartphone app that allows patients to track their use and sends data to doctors so they can view it remotely.
Proteus is working with three pharma companies to test this technology in combination with buprenorphine, naloxone and naltrexone.
A team at the University of Toronto have developed a behind the ear wearable device for opioid treatment. The device, called NSS-2 Bridge, is able to help reduce opiate withdrawal symptoms in adults.
The bridge delivers 3.2 milligrams of a drug called naloxone over 30 minutes when patients feel withdrawal symptoms. It can be used for up to 5 days.
Each bridge costs $80 and is covered by insurance in some states.
The U.S. Food and Drug Administration has approved a behind-the-ear wearable device that reduces withdrawal symptoms caused by stopping opioid use.
The NSS-2 Bridge is a small, non-invasive nerve stimulator device that fits behind the ear and delivers electrical stimulation to the cranial nerves to relieve symptoms of opioid withdrawal.
The FDA said the device can be used for up to five days in patients 18 and older who are undergoing medication assisted therapy for opioid use disorder, as well as those who have stopped taking opioids abruptly and are experiencing withdrawal symptoms.
Symptoms of opioid withdrawal include nausea, vomiting, diarrhea, muscle aches, increased tearing, runny nose and sweating.
“The FDA’s approval of NSS-2 Bridge was based on results from two clinical trials with a total of more than 600 participants,” Dr. William Maisel, acting director of the Office of Device Evaluation at FDA’s Center for Devices and Radiological Health, said in an agency news release. “We are committed to leveraging medical devices as part of our comprehensive public health response to address the opioid crisis.”
The FDA approved NSS-2 Bridge after an expedited review process triggered by the U.S. Opioid Epidemic Act. The device
Researchers at the University of California, Berkeley have developed a behind-the-ear wearable device that could deliver medication to prevent and treat opioid overdose.
The device, which is still in development, consists of a tiny polymer patch and a disposable applicator. The polymer patch contains naloxone, a fast-acting antidote for opioid overdose, and the applicator is used to apply adhesive backing onto the skin behind the ear to hold the patch in place for up to three days. Because the patch is small and discrete, it can be worn with an earbud and won’t interfere with daily activities such as wearing a helmet or swimming.
For people trying to beat opioid addiction, the battle isn’t over once they get clean. A relapse can come at any time, putting them at risk of an overdose.
Probuphine is an implant that continuously releases medication to help reduce cravings for opioids and block the euphoric effects of these drugs. The drug is meant to be used as part of a broader treatment plan, which also includes counseling and behavioral therapy. Probuphine is inserted under the skin on the inside of the upper arm by a healthcare provider during an office visit. It releases medication for six months and then needs to be removed by a doctor.
In clinical trials, patients with opioid dependence were more likely to remain in treatment when using Probuphine compared with placebo (not active drug). While using Probuphine, patients also had fewer positive urine drug tests for opioids than those who received placebo.
What is the Action of a Behind The Ear Wearable Device For The Treatment of Opioid Withdrawal Symptoms?
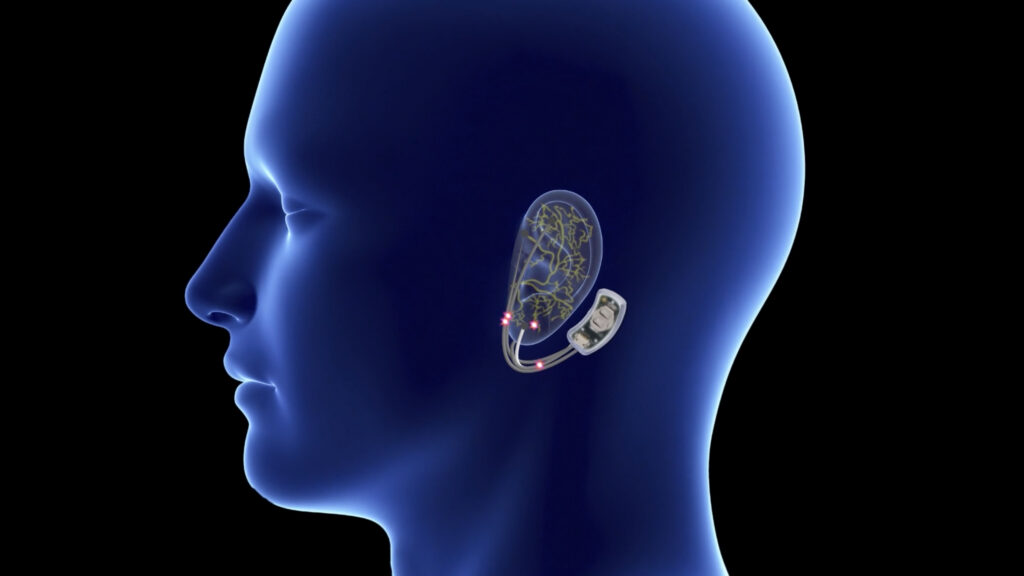
What is the action of a behind the ear wearable device for the treatment of opioid withdrawal symptoms?
The prototype device in question is a behind the ear wearable device that uses an electrical current to stimulate acupressure points behind the ear. These acupressure points are supposed to relieve opioid withdrawal symptoms, but more research is needed to confirm such claims.
Behind-the-ear devices are not new to the medical industry. In fact, they were used to treat smoking cravings in patients who wanted to kick the habit and addiction. However, this new behind-the-ear device is being used for treating opioid withdrawal symptoms instead of smoking cravings, which makes it quite unique in its own right.
The device also does not require any medication or drugs, which means patients can use it without having to worry about potential side effects or health risks associated with drugs and other medications.
There have been some studies done on this particular type of device. One such study found that patients who wore the device for four days reported significant improvements in their withdrawal symptoms compared those who did not wear one at all (i.e., control group).
However, there were only 25 participants involved in this study and they were not randomly assigned either (meaning they were assigned based on
The system is a behind the ear wearable device for the treatment of opioid withdrawal symptoms. The system can also be used to treat substance use disorders. The present invention provides a method of treating an opioid withdrawal symptom in a subject by administering electrical signals at predetermined frequencies and intensities to a branch of the vagus nerve in the ear of the subject, wherein the electrical signals are delivered by a behind the ear wearable device.
The TheraPearl® Opioid Withdrawal System (TheraPearl device) is a behind the ear wearable device that provides targeted localized cold therapy for the treatment of opioid withdrawal symptoms. The cold therapy decreases the severity of symptoms by decreasing inflammation and constricting blood vessels to provide analgesic effect.
The device is a small patch that is placed behind the patient’s ear to deliver the opioid antagonist naltrexone over several days. The drug has been FDA-approved for decades and prevents opioids from binding to their receptors in the brain and other organs, blocking their analgesic effects.
However, the drug must be taken orally every day, which is difficult for many patients given the numerous side effects caused by withdrawal symptoms. Naltrexone also has a short half-life so patients often relapse before the next dose.

The patch is applied behind the ear where it continuously delivers naltrexone through the skin and into the bloodstream. It can be worn for up to seven days and removed by a healthcare professional at any time.
The device utilizes the body’s natural response to changes in light and temperature to alter the body’s internal clock, which is responsible for many of the unpleasant symptoms of withdrawal.
The device is worn behind the ear and uses a proprietary combination of light and temperature therapy to alter the body’s internal clock and help alleviate symptoms.
The device delivers both light therapy and temperature therapy to improve patients’ sleep patterns and offset some of the physical symptoms as well as mental distress associated with withdrawal.
The device delivers both light therapy and cold therapy, which work together to improve sleep patterns, relieve anxiety, and reduce other physical symptoms associated with withdrawal
The device uses electrical signals to stimulate the brain. This stimulation helps to decrease withdrawal symptoms, and is used as a therapy in conjunction with medication assisted treatment.
There are three main types of opioid withdrawal:
Acute Withdrawal Syndrome (AWS) occurs within a few hours after the last dose and may last several days. AWS symptoms include anxiety, insomnia, muscle aches, sweating, runny nose, yawning, and loss of appetite.
Early Withdrawal Syndrome (EWS) lasts for 5–10 days after the last dose. Symptoms include restlessness, chills or goosebumps on skin (“gooseflesh”), stomach cramps, diarrhea, vomiting, muscle aches, sweating, and dilated pupils.
Protracted Withdrawal Syndrome (PWS), also called post-acute withdrawal syndrome (PAWS), can occur months or even years after the last use of opioids. Symptoms include depression or other mood disorders[1] , sleep disruptions[2], fatigue[2], anxiety[2], irritability[2], and craving for the drug.[1]
Other symptoms include agitation,[3] drug craving,[4][5] increased sensitivity to pain,[6] hallucinations,[7] and panic attacks.[8]
What Is Behind The Ear Wearable Device?

Behind the ear wearable device is a wireless headset. It is a very fashionable and comfortable Bluetooth headset, which can be worn behind the ear. It can connect with smartphones through Bluetooth, and it can support voice calling, video chat, music playback and other functions.
Behind the ear wearable device have many advantages:
- Because this Bluetooth Headset is placed behind the ear, it will not feel heavy for a long time.
- The microphone hole of behind the ear wearable device is hidden in the arm, so it is more convenient to make calls without taking off your glasses or headphones.
- This product has two colors of black and white, which are suitable for both men and women, and are suitable for all kinds of scenes such as business occasions, daily sports or travel.
Behind the ear wearable devices are synonymous with Bluetooth headsets, which are small electronic devices that people can wear behind the ears while they are driving and talking on the phone. The person wearing such a device will be able to use their hands and ears freely. The microphone in this device will pick up your voice and send it to the speaker of another device worn by a different person.
Wearables are not just a thing of the future. They are here, they are now and they are taking the world by storm.
The wearables market, which includes health and fitness trackers, smartwatches, smart eyewear and hearables, is projected to grow at an annual rate of 23 percent to reach $25 billion in 2021. That’s a significant increase of 350 percent over 2016.
Let’s take a look at one of the most popular types of wearables for monitoring your health: behind-the-ear wearable devices.
Behind-the-ear hearing aids are larger than in-the-ear or in-the-canal devices and sit outside of the ear.
The size of behind-the-ear devices means that they can be fitted with more technology compared to smaller devices.
Behind the ear hearing aids are suitable for all types of hearing loss and can be used by adults and children.
You’ve seen the behind-the-ear hearing aid, but do you know what it is and how it works?
What Is a Behind-the-Ear Hearing Aid?
The behind-the-ear (BTE) hearing aid is a device that amplifies sounds and directs them into your ear canal, so you can hear better. The device consists of two parts: the part that rests behind the ear and the mold or dome that fits in your ear canal.
How Does It Work?
Sound travels through the tube from the BTE to the mold or dome in your ear. The mold or dome helps direct sound into the ear canal, which improves sound quality. The hearing aid amplifies sounds, so you can hear them better. If you have tinnitus, a BTE can also be fitted with a tinnitus feature to mask the ringing sound.
What Are the Benefits of a Behind-the-Ear Hearing Aid?
You’ll find several benefits to wearing a BTE hearing aid including:
It’s easy to wear and operate
It’s comfortable, because it rests behind your ear rather than in your ear canal
It’s durable, making it ideal for young children who are rough on their devices
When you start working with a cochlear implant, you will be able to hear some sounds, but they may not sound like normal speech. To help your brain learn to understand these new sounds, you will need to practice listening and speaking. It can take up to two years to get the full benefit of your device.
You will likely need special ear molds or headphones to listen to sounds through your implant. In addition, you may need another device called a remote assistant that helps you fine-tune your implant’s settings. You will also receive a computer processor that sits behind the ear and converts sound into electrical signals that are sent to the implant.